mCardia is a context-aware ECG collection system for arrhythmia (irregular heartbeat) screening. This will enable longitudinal contextualized ECG collection in a natural setting and help in optimizing the diagnosis of atrial fibrillation.
The aim of the mCardia system is threefold:
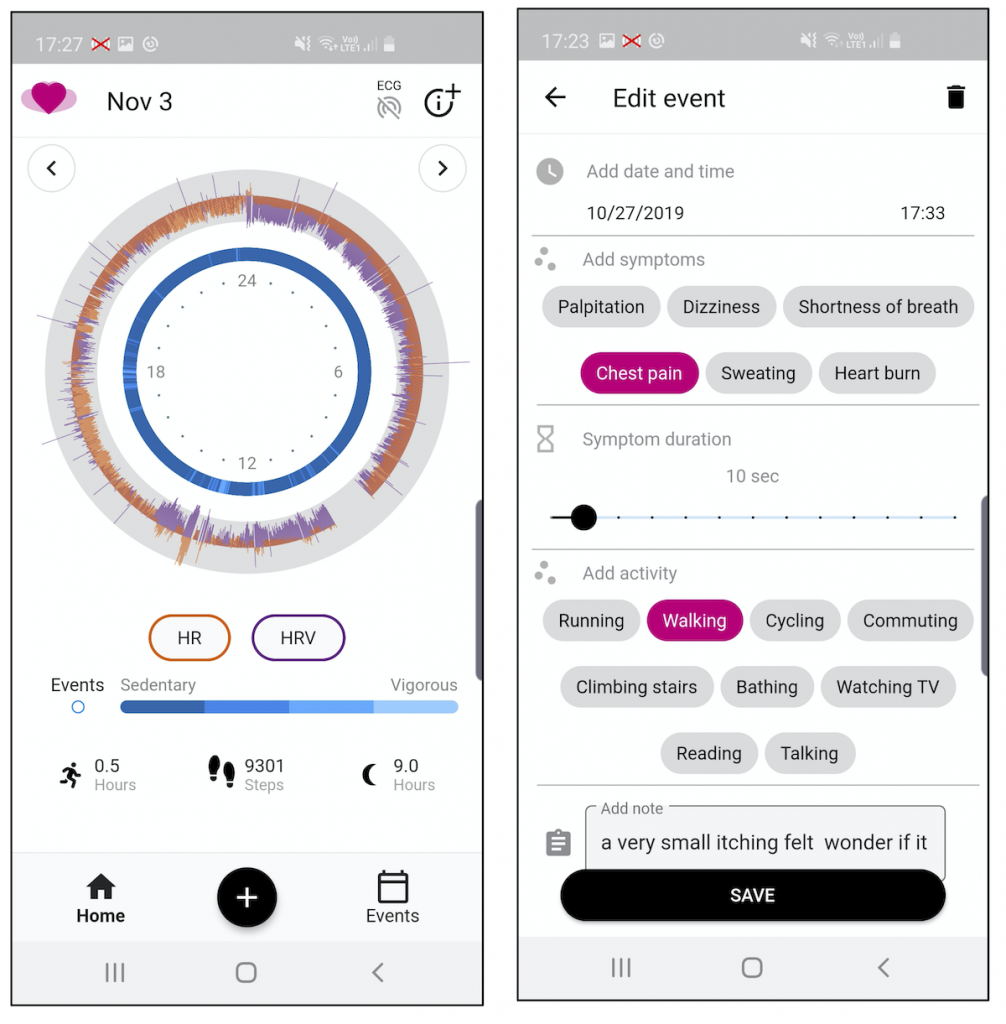
- To collect contextual data (both self-reported and passive) such as unusual symptoms (dizziness, palpation, heartburn, etc.) and their duration, activities (walking, running, exercising, etc.), body position, sleep, food intake and synchronize them with the ECG recordings in order to give a contextualized understanding of the ECG to the cardiologist.
- To reduce recall bias in patient-reported events during ambulatory long-term ECG monitoring.
- To improve doctor and patient communication during longitudinal arrhythmia screening periods (week long) and help the patient in keeping track of unusual symptoms.
mCardia has been developed as a part of the REAFEL (Reaching the Frail Elderly Patient for Optimizing Diagnosis of Atrial Fibrillation) project which is funded by the Innovation Fund Denmark. The mCardia team includes computer scientists, engineers, cardiologists, and user interface designers from the Copenhagen Center for Health Technology (CACHET), the Department of Health Technology at the Technical University of Denmark (DTU), and the Department of Cardiology (Y) at Bispebjerg and Frederiksberg Hospital (BFH). See this page for a summary of the REAFEL project.
Technical use of CARP
The mCardia app is implemented using Flutter but is only available for Android since the attached ECG monitor only works for Android.
The mCardia system make use of most CARP components, including:
- CAMS for mobile sensing of location, activity, step counts, and similar data. It also uses CAMS for integration to the Movisens EcgMove4 device for collecting ECG, HR, HRV, metabolic level, and step count data. This is done via the CAMS Movisens sampling package.
- Research Package for collection of informed consent.
All data is uploaded and managed in CAWS hosted by CACHET.
References
- Kumar, D., Maharjan, R., Maxhuni, A., Dominguez, H., Frølich, A., & Bardram, J. E. (2022). mCardia: A Context-Aware ECG Collection System for Ambulatory Arrhythmia Screening. ACM Transactions on Computing for Healthcare (HEALTH), 3(2), 1-28.